Health Promotion Perspectives, 10(3), 180-191; DOI:10.34172/hpp.2020.31
Systematic Review
Psychological interventions for depression and anxiety: a systematic review and meta-analysis of Iranian chronic pain trials
Gholamreza Jandaghi1, Manijeh Firoozi2, Ali Zia-Tohidi3,*
1
Department of Management, Faculty of Management and Accounting, Farabi Campus, University of Tehran, Qom
2
Department of Psychology, Faculty of Psychology and Educational Sciences, University of Tehran, Tehran, Iran
3
Faculty of Management and Accounting, Farabi Campus, University of Tehran, Qom
Email: towhidi@ut.ac.ir
© 2020 The Author(s). This is an open access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Background: Chronic pain is commonly associated with anxiety and depression, making it more challenging to be managed. Psychological interventions are suggested for such complicated issues which are well evident in the United States and Europe. However, generalizing the evidence to Iranian population – as a Middle Eastern society – might be questionable. We aimed to synthesize our evidence on the effectiveness of these interventions among Iranian populations.
Methods: This was a systematic review and meta-analysis. Persian and English literature were searched through Iran-doc, Elm-net, and PubMed until March 2019 using the following terms (or its Persian synonyms): chronic pain; persistent pain; chronic fatigue; fibromyalgia; neuropath*; LBP; irritable bowel; CFS; psycho*; cogniti*; acceptance; meaning; mindfulness; relaxation; biopsychosocial; rehabilitation; educat*. Eligible trials were randomized trials that evaluated the effectiveness of psychological interventions on Iranian adults with chronic pain. No setting restriction was considered. Risk of bias for each trial was assessed, and the random-effect model was used to pool summary effect across trials.
Results: In all 30 eligible RCTs, the risk of bias for randomization was low except for one study. The pooled standardized mean difference (SMD) for depression and anxiety were 1.33 (95%CI: -1.42 to -0.68) and 1.25 (95% CI: -1.55 to -0.96), respectively.
Conclusion: This study suggests that psychological interventions are highly effective in reducing depression and anxiety in Iranian patients with chronic pain, compared to what observed in theU.S. and European studies. However, there are still some methodological issues to be addressed. Future research should focus on high-quality trials with considerations on the methodological issues reported in the present study.
Keywords: Depression, Anxiety, Chronic pain, Psychotherapy, Meta-analysis, Iran
Introduction
Treating or well managing chronic pain (CP) has been considered as an unresolved issue for decades. Various medical and psychological treatments are shown to be slightly effective in relieving pain.1 On the other hand, CP commonly co-occurs with anxiety and depression,2-4 which adds more complications to the health status of the patients. According to a country-level survey in Iran, depression is the leading and the second cause of years lost due to disability in women and men, respectively.5 The issue becomes more dramatic when we realize that low back pain is the other leading cause (first cause in men and the second in women). Beside depression and anxiety as two major risk factors for suicide ideation and attempt,6 CP is also considered as one of the main risk factors for suicide, as reported by the World Health Organization (WHO).7
The clinical recommendations suggest that when these conditions occur concomitantly, they should be treated concurrently8; as CP has complicated relationships with anxiety and depression which necessitate simultaneous treatment:
Depression and pain share neurological pathways as well as psychological factors (e.g., catastrophizing, feeling of loss, behavioral avoidance), and respond to same treatments, both pharmacological (e.g., TCAs, SSRIs) and psychological (e.g., CBT).9
Evidence suggests that anxiety and depression could exacerbate pain intensity, facilitate the transition of the acute to CP,10 or even have a causal effect on the onset of pain, especially in the case of anxiety. In fact, there is evidence that in most cases of co-occurrence, anxiety precedes the onset of CP, unlike depression which usually occurs after the development of CP.11
There is longitudinal evidence that high level of pain interference at baseline could result in poor treatment outcomes on anxiety and depression.12,13
Improvement in depression and anxiety during treatment could improve pain outcomes, such as pain intensity/interference and disability, as well.14
Due to these interactions, and more generally due to the biopsychosocial nature of CP, related organizations in the United States have recently recommended a multidimensional framework for the diagnosis of CP conditions that incorporate social and psychological aspects of the issue —10,15 analogous to the multiaxial framework used in DSM-3 and 4.
To address anxiety and depression in patients with CP, researchers worldwide have conducted hundreds of RCTs (especially on psychological treatments as the interest of this research), which has been summarized in numerous reviews and meta-analyses.16-21 However, the evidence is mainly based on the trials conducted in the United States and Western European countries, and though it might not be generalizable to other regions around the globe, especially to the regions with considerable cultural differences–such as the Middle East. In Hilton et al,20 30 out of all included trials were conducted in the United States and Europe, and only 4 were performed in the Middle East; and In Bernardy et al,21 28 out of all total 29 included trials were conducted either in the United States or Europe.
Culture, chronic pain, and psychotherapy
Our skepticism about the generalizability of the American and European trials to a Middle Eastern population such as Iranian community is based on the fact that pain is a biopsychosocial phenomenon.10,22,23 It acts and responds differently in various cultural contexts. Patients’ beliefs about pain and its consequences, their behavioral response to pain, as well as their expectations, views, reactions, and perceived supports could change the way that pain affects them. (for a review on cultural differences in pain perception see Pillay et al24).
On the other hand, almost all well-known psychotherapies have been developed in American and European cultures, by theoreticians who grew within those environments, and through working with subjects from those cultures. Such theories may not fit well to the situations happened in other cultures.25
Also, concerning CP treatments, the main issue in most psychological interventions for CP is probably coping with pain (stress), which has a different pattern across various cultures. For instance, there is evidence that people from collectivistic cultures use more emotion-focused strategies than they do in individualistic cultures. Also, some coping strategies are helpful in some cultures and not in others.26,27
Our objective in this study was to evaluate and synthesize Iranian randomized clinical trials that investigated the efficacy of psychological interventions on anxiety and depression in adults with CP compared to usual care or waitlist. Then, we compared our results with other meta-analytical results, and tried to stress the differences between them, not only concerning the effect sizes (ESs) but also other aspects of trials, such as methodology.
Materials and Methods
This was a systematic review and meta-analysis, which was reported based on the PRISMA guideline.
Eligibility criteria
Participants and settings
Eligible participants were Iranian adults diagnosed with CP (defined as the pain that lasts for more than three months). Studies on the patients with chronic cancer pain and CP due to multiple sclerosis (As both diseases have a high level of effect on anxiety and depression, regardless of pain) were excluded. Also, the trials with less than ten participants per group were excluded. No setting restriction was applied.
Interventions
All psychological interventions with all formats of delivery (e.g., individual therapy, group therapy) were eligible. To reduce heterogeneity, the interventions with merely medical education and the intervention courses with less than four treatment sessions were excluded.
Type of control
Control groups included treatment as usual (TAU; or usual care), waitlist and psychological placebo. Accordingly, trials with other active controls were excluded (i.e., the trials within which two psychological approaches were compared).
Outcome measures
Studies that reported outcomes on depression or anxiety were included.
Study design
Only RCTs that reported as either journal articles or theses were included.
Information sources, search strategy, and study selection
We searched Iran-doc, Elm-net, and PubMed (until March 2019) for eligible trials using the following terms (or its Persian synonyms): chronic pain; persistent pain; chronic fatigue; fibromyalgia; neuropath*; LBP; irritable bowel; CFS; psycho*; cogniti*; acceptance; meaning; mindfulness; relaxation; biopsychosocial; rehabilitation; educat*. As such a comprehensive search strategy in PubMed resulted in about 35 000 records of which only a small proportion conducted in Iran we imposed the following additional terms: Iran; Iranian; or Persian. Titles, abstracts, and then the whole texts were screened (flow diagram of selection process presented in Figure 1).
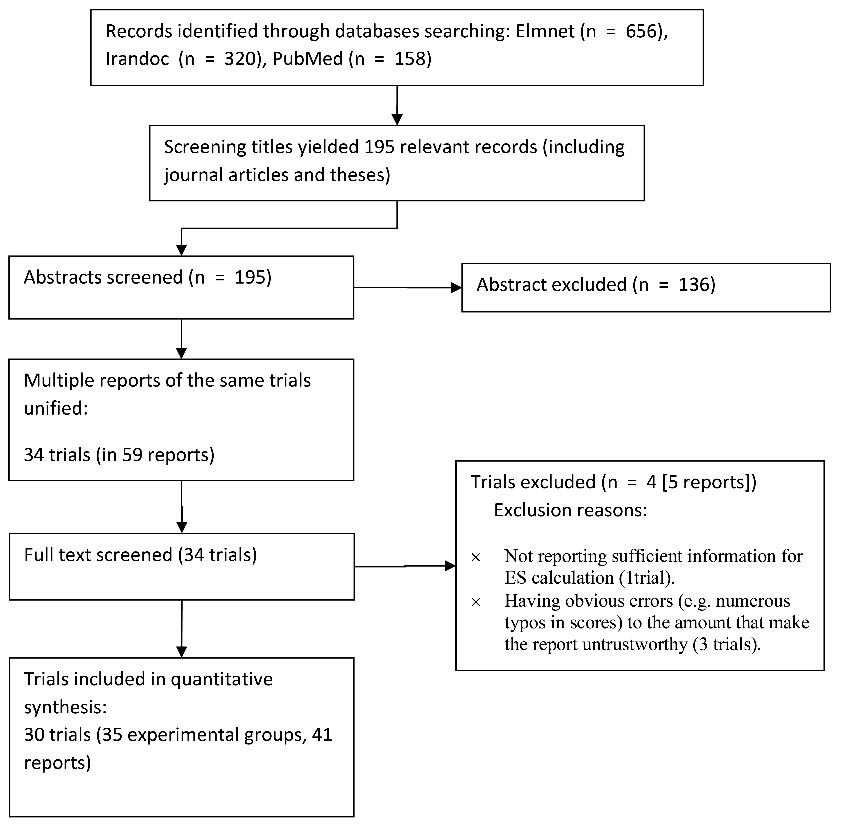
Figure 1. Flow diagram of study selection process.
Assessing risk of bias in individual trials
We assessed the risk of bias at the study level, based on the Cochrane collaboration guidelines in four domains: sequence generation, allocation concealment, incomplete outcome data, and selective outcome reporting.28 Blindness was not assessed in this study, because blinding the therapists and the patients are usually not applicable in psychological treatments. Also, in all included studies data were collected through self-report. So, blinding the outcome assessors was inapplicable.
Data preparation and synthesis methods
Calculating within study effect sizes
We used standardized mean difference (SMD) to measure ES, as the unstandardized mean difference was not appropriate due to different measurement scales across the trials. To do so, we adjusted the baseline scores, as the most of included trials had small samples, and in the studies with small sample size baseline differences could substantially affect the results. The preferred approach to account for baseline scores is the use of information from analysis of covariance applied to estimate adjusted ESs.29,30 However, only a few studies reported such information. So, we used Morris formula to calculate SMD based on pre-test and post-test means and pre-test standard deviations, which gives more precise estimates from ESs than common formula based on post-test scores.31 Hedges correction for small studies applied to all ESs.
Addressing multi-arm trials
In multi-arm trials that compare two or more experimental arms to one shared control group, a somehow common mistake is to repeat the control group in the analysis to compare with each treatment arm. This measure may put excessive weight on these trials (by double-counting the control) and ignore the correlation between ESs. To deal with this issue, we combined the experimental arms (as recommended by the Cochrane32). But, this approach does not allow to conduct subgroup analysis based on treatment type. Therefore, we used another solution—to split the shared control group.
Synthesis method
Meta-analysis was conducted using a random-effect model with restricted maximum likelihood, as the estimator of true heterogeneity, which seems to provide us with less biased estimates than other methods.33
Additional analyses
For each analysis, we checked for potential outliers (based on studentized residuals) and influential cases (based on an index analogous to cook’s distance in meta-analytic context34). We did not conduct any analysis to detect publication bias. Because fail safe-N has several problems and is not recommended to be used. Also, using funnel plot and the related tests are appropriate only if there were considerable differences between trials in terms of their sample size, and at least some of them have large samples which raise their publication chance, even if their results are not statistically significant. This logical basis was not true in the context of our study (for more details on these methods, see Jin et al35). Subgroup analyses were conducted based on three pre-specified factors: treatment type, researcher’s education (Ph.D. or M.A.), and pain type.
Statistical software
All analyses were conducted in R using the package “metafor”,36 except for calculating SMD from ANCOVA which was conducted in the package “compute.es”.37
Results
Our search and screening resulted in 30 eligible trials, including 35 experimental groups, reported in 54 papers/theses (flow diagram of the selection process is presented in Figure 1). The total sample size was 1021 (with an average of 34 participants per trial). The most common condition across trials was irritable bowel syndrome (IBS) (43%), followed by low back pain (n = 7 [23%]). Female patients constituted 86% of the total sample, with 18 trials conducted only on female patients (60% of trials). One trial was conducted on male patients, and the others included participants from both men and women. The majority of trials used either cognitive-behavioral or third wave treatments. Twelve trials conducted follow up assessment with an average period of 8.6 weeks. The characteristics of the included studies are presented in Table 1.
|
Table 1. Characteristics and risk of bias for each included trial
|
|
Study
|
References
|
Follow up (week)
|
Pain type
|
Intervention (delivery, sessions)
|
Outcomemeasures
a
|
Sample size
|
Female %
|
Risk of bias
|
|
|
|
|
|
|
|
|
Sequence generation
|
Allocation concealment
|
Incomplete data
|
Selective report
|
| Alighias 2013 |
41,42
|
12 |
LBP |
Psychological education (group, 4) |
DASS-21 |
84 |
100% |
? |
? |
√ |
√ |
| Anvari 2013 |
43
|
8 |
CP |
ACT (group, 8) |
DASS-21 |
17 |
0% |
? |
? |
O |
√ |
| Arvand 2017 |
44
|
- |
IBS |
Music therapy (group, 10) |
BAI |
69 |
100% |
? |
? |
? |
? |
| Asadollahi 2013 |
45
|
8 |
IBS |
(1) MBCT (group, 8)
(2) spirituality therapy (group, 8) |
SCL-90 |
30 |
100% |
? |
? |
? |
√ |
| Esmaeelzadeh 2017 |
46,47
|
- |
IBS |
Emotional intelligence training (group, 12) |
BAI |
36 |
Unclear |
? |
? |
√ |
? |
| Adarvishi 2016 |
48
|
- |
IBS |
Problem solving training (group, 8) |
STAIb |
46 |
65% |
? |
? |
√ |
? |
| Foroodastan 2013 |
49
|
- |
MSP |
(1) CBT (individual, 8)
(2) quality of life therapy (individual, 8) |
DASS-42 |
30 |
100% |
? |
? |
√ |
O |
| Poormohseni 2017 |
50
|
- |
IBS |
MBCT (group, 8) |
GHQ-28 |
40 |
65% |
? |
? |
√ |
? |
| Golchin 2011 |
51,52
|
- |
LBP |
CBT (group, 12) |
DASS-21 |
30 |
100% |
? |
? |
√ |
√ |
| Hazrati 2007 |
53,54
|
12 |
IBS |
Bensons' relaxation (audio tape, 3 months daily) |
STAI |
30 |
73% |
O |
? |
O |
? |
| Haghaegh 2008 |
55
|
8 |
IBS |
CBT (group, 8) |
BDI |
28 |
58% |
? |
? |
O |
√ |
| Irandoost 2014 |
56
|
8 |
LBP |
ACT (group, 8) |
HADS
CES-D |
40 |
100% |
? |
? |
√ |
O |
| Jokar 2009 |
57
|
4 |
RA |
Cognitive behavioral stress management (group, 10) |
DASS-42 |
16 |
100% |
? |
? |
O |
√ |
| Khoshsorour 2018 |
58
|
- |
IBS |
Neurofeedback (individual, 10) |
STAI |
30 |
100% |
? |
? |
√ |
? |
| Soltanian 2014 |
59
|
- |
RA |
CBT (group, 8) |
DASS-21 |
20 |
100% |
? |
? |
? |
√ |
| Sabour 2016 |
60
|
4 |
CP |
ACT (group, 8) |
DASS-21 |
16 |
100% |
? |
? |
O |
O |
| Tabatabaie 2014 |
61
|
- |
IBS |
Meta cognitive therapy (group, 8) |
HADS |
21 |
100% |
? |
? |
√ |
√ |
| Kamkar 2011 |
62
|
12 |
IBS |
Cognitive behavioral stress management (group, 8) |
BAI |
42 |
48% |
? |
? |
? |
√ |
| Ma'soomian 2013 |
63,64
|
- |
LBP |
MBSR (unclear, 8) |
DASS-21 |
18 |
100% |
? |
? |
O |
O |
| Rahimian Boogar 2012 |
65
|
16 |
LBP |
CBT+pain management (group, 8) |
DASS-42 |
35 |
48% |
√ |
√ |
? |
√ |
| Salayani 2015 |
66,67
|
3 |
CP |
(1) CBT (group, 8)
(2) CBT+pain management (group, 8) |
DASS-21 |
30 |
100% |
? |
? |
√ |
√ |
| Shojaie 2016 |
68
|
- |
CP |
The transtheoretical model (stages of change) (group, 8) |
BDI |
30 |
100% |
? |
? |
√ |
√ |
| Mohammadi 2017 |
69
|
- |
IBS |
MBCT (group, 8) |
BAI |
40 |
Unclear |
? |
? |
√ |
√ |
| Mahvi shirazi 2008 |
70,71
|
- |
IBS |
CBT (unclear, 8) |
SCL-90 |
50 |
59% |
? |
? |
√ |
√ |
| Vakili 2009 |
72,73
|
8 |
LBP |
CBT (group, 8) |
SCL-90 |
26 |
100% |
√ |
? |
? |
√ |
| Yeganeh 2015 |
74
|
- |
RA and lupus |
MBSR (group, 8) |
DASS-21 |
30 |
100% |
? |
? |
√ |
√ |
| Yusof Zadeh 2017 |
75,76
|
8 |
LBP |
(1) CBT (individual, 6)
(2) schema therapy (individual, 6) |
DASS-21 |
35 |
74% |
√ |
? |
O |
? |
| Mo'tamedi 2012 |
77
|
- |
Headache |
ACT (group, 8) |
STAI-trait |
26 |
100% |
? |
? |
O |
O |
| Moghtadaie 2012 |
78,79
|
8 |
IBS |
MBCT (group, 8) |
BSI |
14 |
100% |
? |
? |
O |
√ |
| Nazemi Ardakani 2016 |
80,81
|
- |
RA |
(1) Cognitive behavioral stress management (group, 8)
(2) Islamic spiritual therapy (group, 8) |
DASS-21 |
62 |
Mostly |
? |
? |
√ |
√ |
|
Abbreviations: LBP = low back pain; CP = chronic pain; IBS = irritable bowel syndrome; MSP = musculoskeletal pain; RA = rheumatoid arthritis; ACT = acceptance and commitment therapy; MBCT = mindfulness-based cognitive therapy; CBT = cognitive behavioral therapy; MBSR = mindfulness-based stress reduction; DASS = depression, anxiety, and stress scale; BAI = Beck anxiety inventory; SCL = symptom check list; STAI = state-trait anxiety inventory; GHQ = general health questionnaire; BDI= Beck depression inventory; HADS = hospital anxiety and depression scale; BSI = brief symptom inventory.
a It only includes outcome measures used in this meta-analysis. In multifactor scales (e.g., SCL-90, GHQ-28), only scores from anxiety and/or depression subscales were used.
b In this measure, we used the state subscale, as it is sensitive to change, and which is comparable to other anxiety scales.
√ = low risk of bias; O = high risk of bias; ? = unclear risk of bias.
|
Risk of bias in included trials
The risk of bias due to randomization processes (i.e., sequence generation and allocation concealment) was high or unclear in all trials with one exception. Half of the trials were at high or unclear risk of incomplete cases, and less than half were in selective outcome reporting (see Table 1).
Depression
At post-treatment, pooled SMD from 23 trials was -1.42 (95% CI: -1.76 to -1.09), with significant heterogeneity between trials (τ2 = 0.47, Q(df = 22) = 80.1, PQ < 0.001, I2 = 74%). Additional investigation detected one outlier with an unexpectedly huge ES (d = -6.18, studentized deleted residual = -3.64). This study had huge ESs in anxiety, as well (Cohen’s d in anxiety was more than 3). So, we removed it from further analyses in all outcomes. The removing of this study resulted in a slight reduction in ES (SMD = -1.33), but heterogeneity remained significant (τ2 = 0.34, Q(df = 22) = 62.8, PQ < 0.001, I2 = 68%). Among our pre-specified potential moderators, intervention type significantly moderated the effect, explaining 22% of the heterogeneity (Table 2), but the residual heterogeneity was still significant (PQ < 0.05). More details about subgroup analyses are presented in Table 2 and the forest plot of individual ESs within each study are presented in Figure 2.
|
Table 2. Summary of meta-analytical results and subgroup analyses
|
|
Factor
|
Condition
|
k
|
N
|
SMD
|
95% CI
|
Test for moderator
|
Depression
(Post-treatment) |
|
Total
|
22
|
684
|
-1.33
|
-1.64, -1.03
|
-
|
|
Treatment type |
Cognitive behavioral |
11 |
277 |
-0.90 |
-1.27, -0.54 |
Q (df = 2) = 5.84, P = 0.05, R2 = 23%
|
|
|
Mindfulness-based |
11 |
301 |
-1.49 |
-1.86, -1.12 |
|
|
|
Others |
5 |
106 |
-0.86 |
-1.44, -0.28 |
|
|
Pain type |
MSC |
15 |
419 |
-1.52 |
-1.89, -1.15 |
Q (df = 1) = 2.55, P = 0.11, R2 = 9%
|
|
|
IBS |
7 |
265 |
-1.02 |
-1.50, -0.55 |
|
|
Researchers education |
MA |
14 |
350 |
-1.46 |
-1.85, -1.07 |
Q (df = 1) = 0.98, P = 0.32, R2 = 0%
|
|
|
PHD |
8 |
334 |
-1.15 |
-1.62, -0.67 |
|
| Depression (follow up) |
|
Total
|
10
|
273
|
-1.05
|
-1.42, -0.68
|
-
|
|
Treatment type |
Cognitive behavioral |
6 |
166 |
-1.01 |
-1.53, -0.49 |
Q (df = 2) = 0.17, P = 0.92, R2 = 0%
|
|
|
Mindfulness-based |
4 |
62 |
-0.94 |
-1.67, -0.21 |
|
|
|
Others |
3 |
45 |
-0.80 |
-1.65, +0.05 |
|
|
Pain type |
MSP |
6 |
159 |
-1.35 |
-1.70, -1.00 |
Q (df = 1 ) = 8.6, P = 0.003, R2 = 100%
|
|
|
IBS |
4 |
114 |
-0.57 |
-0.96, -0.18 |
|
|
|
MA |
6 |
131 |
-0.85 |
-1.33, -0.38 |
Q (df = 1) = 1.43, P = 0.23, R2 = 15%
|
|
|
PHD |
4 |
142 |
-1.29 |
-1.82, -0.76 |
|
| Anxiety (post-treatment) |
|
Total
|
20
|
687
|
-1.25
|
-1.55, -0.96
|
-
|
|
Treatment type |
Cognitive behavioral |
7 |
168 |
-0.96 |
-1.49, -0.43 |
Q (df = 2) = 1.23, P = 0.54, R2 = 0%
|
|
|
Mindfulness-based |
10 |
293 |
-1.34 |
-1.77, -0.91 |
|
|
|
Others |
7 |
226 |
-1.15 |
-1.65, -0.65 |
|
|
Pain type |
MSP |
8 |
259 |
-1.04 |
-1.48, -0.61 |
Q (df = 2) = 5.18, P = 0.07, R2 = 19%
|
|
|
IBS |
11 |
402 |
-1.29 |
-1.66, -0.92 |
|
|
|
Headache |
1 |
26 |
-2.78 |
-4.24, -1.33 |
|
|
Researchers education |
MA |
13 |
397 |
-1.39 |
-1.75, -1.02 |
Q (df = 1) = 1.35, P = 0.24, R2 = 7%
|
|
|
PHD |
7 |
360 |
-1.03 |
-1.50, -0.55 |
|
| Anxiety (follow up) |
|
Total
|
6
|
217
|
-0.67
|
-0.94, -0.39
|
|
|
Treatment type |
Cognitive behavioral |
2 |
57 |
-0.48 |
-1.01, +0.06 |
Q (df = 2) = 2.69, P = 0.26, R2 = 0%
|
|
|
Mindfulness-based |
3 |
46 |
-1.13 |
-1.77, -0.47 |
|
|
|
Others |
3 |
114 |
-0.56 |
-0.94, -0.18 |
|
|
Pain type |
MSP |
3 |
131 |
-0.80 |
-1.42, -0.17 |
Q (df = 1) = 0.01, P = 0.95, R2 = 0%
|
|
|
IBS |
3 |
86 |
-0.77 |
-1.45, -0.09 |
|
|
Researchers education |
MA |
4 |
145 |
-0.92 |
-1.50, -0.35 |
Q (df = 1) = 0.63, P = 0.43, R2 = 0%
|
|
|
PHD |
2 |
72 |
-0.54 |
-1.29, +0.20 |
|
|
Abbreviations: MSP = musculoskeletal pain; IBS = irritable bowel syndrome.
|
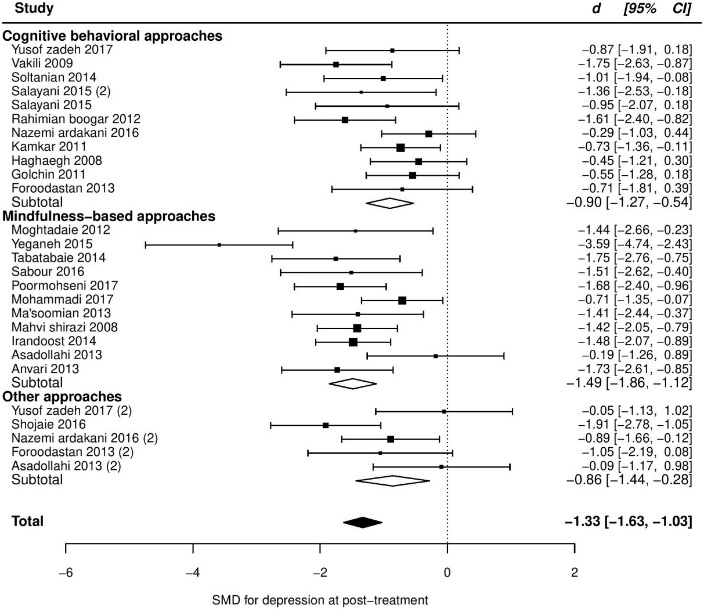
Figure 2. Forest plot of group differences in depression at post-treatment
At follow up, pooled SMD from 10 trials was -1.05 (95% CI: -1.42, -0.68), with significant heterogeneity between trials (τ2 = 0.17, Q(df = 10) = 17.6, PQ < 0.04, I2 = 49%). One study had a positive ES (d = +0.06, indicating negative effect on depression; studentized deleted residual = +2.69). After removing this study, ES increased slightly (SMD = -1.16) and heterogeneity reduced to a non-significant level (τ2 = 0.03, Q(df = 9) = 8.7, PQ < 0.35, I2 = 16%). However, the data point seemed to be valid, and though it may not be a good idea to be deleted, unless as a part of sensitivity analysis. So, we did not remove this study from other analyses. Retaining this study in the model and adding pain type as a moderator also explained almost all observed heterogeneity. Given this subgroup analysis, the patients with musculoskeletal pain were found to significantly experience more improvement than the patients with IBS. More details are presented in Table 2 and the forest plot of individual ESs is presented in Figure 3.
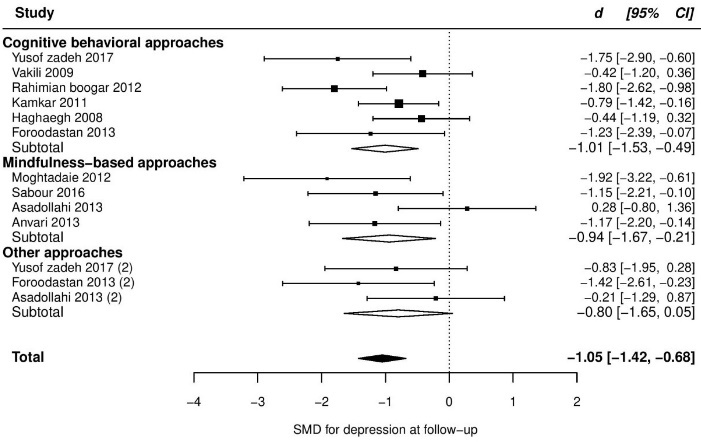
Figure 3. Forest plot of group differences in depression at follow-up.
Anxiety
Twenty trials reported post score on anxiety, which resulted in a large and significant, but very heterogeneous ES (SMD = -1.55, 95% CI [-2.13 to -0.97], τ2 = 1.64, Q(df = 20) = 122.8, PQ < 0.001, I2 = 92%). Additional investigation revealed an outlier with an unusual effect and high influence on the fitted model (d = -8.55, studentized deleted residual = -6.65, cook’s d = 1.00). After removing this study, we found a slight reduction in the pooled ES (SMD = -1.25, 95% CI, -1.55 to -0.96), but heterogeneity remained moderate and highly significant (τ2 = 0.3, Q(df = 19) = 62.1, PQ < 0.001, I2 = 69%). Between our pre-specified moderators, pain type showed a marginally significant moderation effect explaining 19% of the variation. But this moderation effect was, in fact, a function of one category containing one study with a high ES (see Table 2), even though residual heterogeneity was still highly significant (PQ < 001). More details are presented in Table 2 and the forest plot of individual ESs is presented in Figure 4.
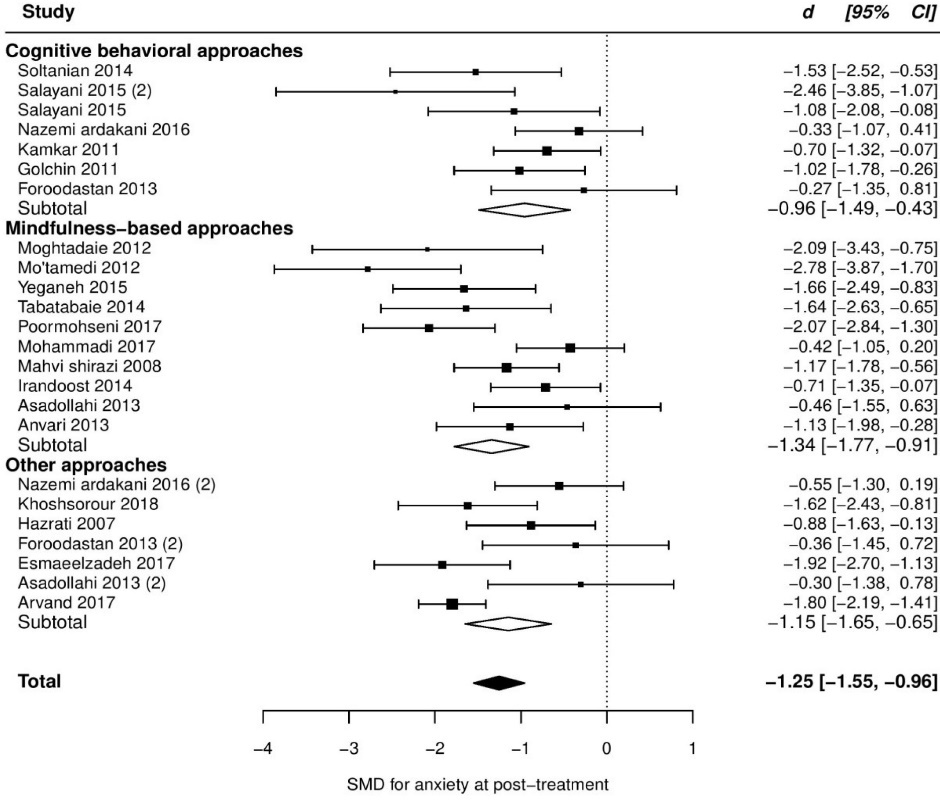
Figure 4. Forest plot of group differences in anxiety at post-treatment.
At follow up, pooled SMD from 7 trials was very large and significant (SMD = -1.34, 95% CI [-2.32 to -0.36]), with very high heterogeneity between ESs (τ2 = 1.52, Q(df = 6) = 37.2, PQ < 0.001, I2 = 91%), which was due to an outlier with an unusual ES (d = -4.3, studentized deleted residual = -5.34). After removing this data-point, pooled ES dropped to one half (SMD = -0.67, -0.94, -0.39), and heterogeneity declined to a non-significant level (τ2 < 0.01, Q(df = 5) = 8.6, PQ = 0.13, I2 = 0%). Comparing our follow up to post-treatment ES, we observed a substantial decline in summary effect. Such a decline may be merely due to the possibility that the studies conducted follow up assessment, in general, reported lower ESs, not only at follow up but also at post-treatment. To test this hypothesis, we ran a meta-analysis on the post-treatment scores of the trials with follow up assessment. Results showed these trials with smaller pooled ES at post-treatment as well (SMD at post-treatment = -0.74, 95% CI [-1.12 to -0.36])- approximately equal to their follow up ES. More details are presented in Table 2 and the forest plot of individual ESs is presented in Figure 5.
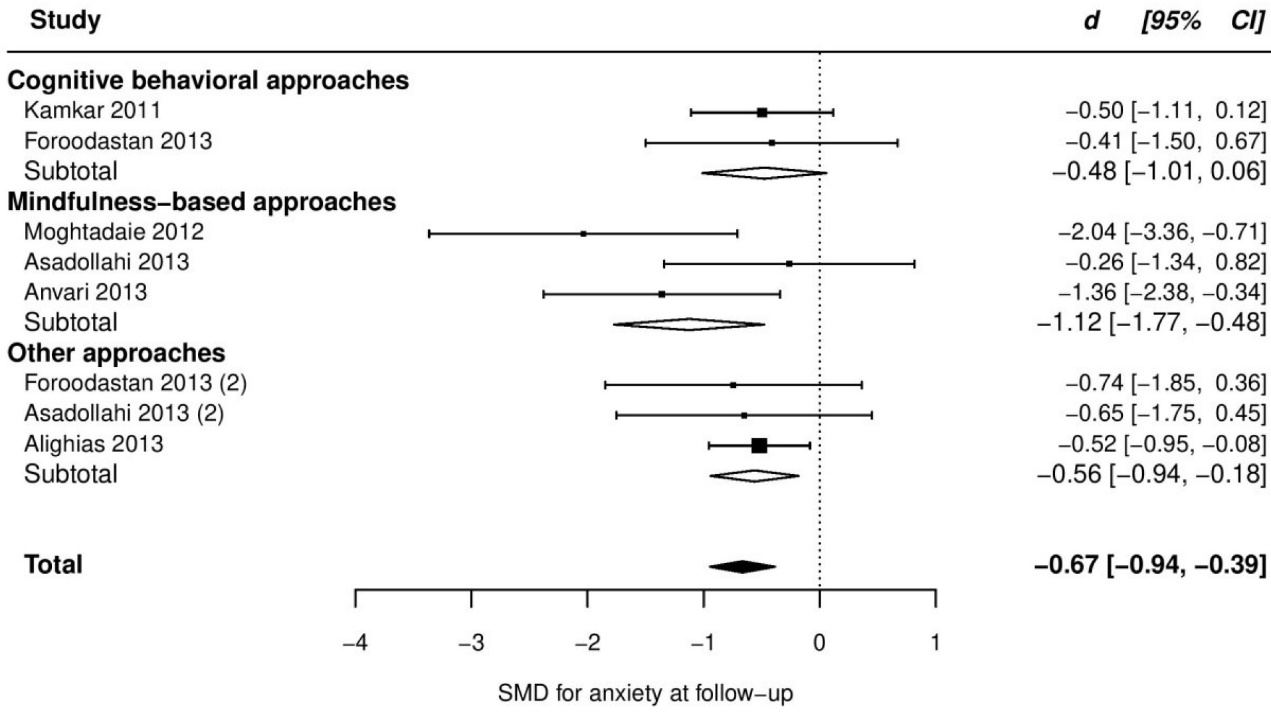
Figure 5. Forest plot of group differences in anxiety at follow-up.
Discussion
Our results suggested that psychological interventions improve both depression and anxiety in Iranian patients with CP, with approximately equal and large ESs. These effects, however, seem to vary across situations and may decline slightly over the time of post-interventions. Subgroup analyses revealed a possible superiority of mindfulness-based treatments over other approaches. Also, patients with musculoskeletal pain may respond better to psychotherapies than patients with IBS, though both groups gain large benefits from these treatments. Other subgroup analyses, based on the researcher’s education level, suggested that the trials conducted by MA researchers, compared to the other interventionists, might result in better effects. It is noteworthy that the results of subgroup analyses should be considered as exploratory findings,29,38 which need to be tested directly in original studies. Also, generalizing our results to male patients might be questionable, as they comprised only about 15% of our total sample size. Although, given this gender distribution, we could be confident in the efficacy of psychotherapies in women.
Comparing our results to those reported in previous meta-analyses (which were mainly conducted in the United States and Europe) we identified substantial differences in the ESs, both in depression and anxiety. Those meta-analyses mostly reported small to medium-sized effects for psychological interventions on depression, and almost the same on anxiety (ESs from 8 previous meta-analyses were presented in Table 3). However, in our included trials the estimated ESs were large, both for depression and anxiety.
|
Table 3. Reported effect sizes in previous non-Iranian meta-analyses and possible characteristics which might explain the difference
|
|
Study (Reference)
|
SMD for depression:
Post-treatment (follow up)
|
SMD for anxiety: Post-treatment (follow up)
|
Characteristics which might contribute in the magnitude of SMDs
|
| Current study |
-1.33 (-1.05) |
-1.25 (-0.67) |
Trials with adequate randomization = 3%
Mean sample across included trials = 34 |
| Morley et al 1999 16 |
-0.36 |
- |
Trials with clear randomization details = 16%
Mean of the sample size across included trials = 57
Objective outcomes (not self-reports) = 23% |
| Williams et al 201217 |
CBT: -0.38 (-0.26)
BT: -0.53 (-0.65) |
- |
Trials with adequate randomization = 24%
Mean sample across included trials = 114 |
| Hoffman et al 200718 |
-0.31 |
- |
Mean sample = 79
Exclusive to chronic low back pain |
| Veehof et at 201619 |
-0.43 (-0.50) |
-0.51 (-0.57) |
Mean sample = 51
Only mindfulness and acceptance-based treatments |
| Hilton et al 201620 |
-0.15 |
- |
Mean sample = 92
Only mindfulness-based treatments |
Bernardy et al
201721 |
-0.43 (-0.48) |
- |
28% with low risk in randomization
Mean sample = 86
Only CBT for fibromyalgia |
| Haugmark et al 201982 |
-0.49 (-0.48) |
-0.37 (-0.34) |
44% with low risk for randomization
Mean sample = 83
Only mindfulness and acceptance-based treatments for fibromyalgia |
| Dixon et al 200783 |
-0.21 |
-0.28 |
Mean sample = 126
Only patients with arthritis |
|
Abbreviations: CBT = cognitive behavioral therapy, BT = behavioral therapy.
|
These differences could be attributable to at least three possible reasons:
First , differences among populations in pain perception and its related constructs and also their reactions to psychotherapies could affect therapeutic outcomes. However, investigating these factors is beyond the scope of our study, and requires cross-cultural investigations. This factor can moderate the true underlying ES of a given intervention on different populations.
Second , differences in the characteristics of included trials could have also changed the magnitude of the observed effects. Obviously, this factor moderates the estimated ES only, and not the true underlying effect. Several well-known factors could elevate the ES. In Table 3, we compared our included trials to the trials included in previous meta-analyses in terms of these factors. The randomization process (including random sequence generation and allocation concealment) may be noticed as an example. Empirical evidence suggests that the trials with adequate randomization tend to produce smaller ESs,28 particularly in subjective outcomes,39 which is the case in our study. In the included trials of our study, only one trial (3%) had low risk of bias due to randomization. Sample size may be considered as another factor. The correlation between sample size and ES was estimated to be as high as -0.52 in tests of mean difference,40 indicating that studies with larger samples produce smaller ESs. The most of trials included in our meta-analysis had relatively small sample sizes (on average 34 participants per trial), which are much smaller than those reported in the trials included in previous meta-analyses (see Table 3).
Third , cultural differences may affect the results of a trial, not due to the population’s differences in response to treatment, but as a result of the sampling process of the trial. Among cultures with a common bias towards psychotherapies, or populations who view pain as a purely physical phenomenon, the participants who join psychotherapy trials may have special characteristics, such as openness. In fact, research samples in pain trials in some cultures may not be a representative of the patients with CP in that culture. So, we may consider this factor as culture’s effect on the external validity of trials.
However, we also found considerable differences in the trials’ characteristics between Iranian and non-Iranian studies (mainly from the United States and Europe).It is difficult to assume that these factors are responsible for all the observed differences between the ESs. Of course, it is possible that the underlying ES to be truly different. Consequently, if the Iranian CP population or other similar populations (e.g., most countries in the Middle East) respond better to psychological treatments, it would be a great opportunity for improving health, particularly in such a pervasive health problem. To that end, conducting large trials with rigorous methodology and clear reports (which facilitate replication) is probably the first step.
Conclusion
Our results suggested that psychological interventions are highly effective in reducing anxiety and depression among Iranian patients with CP; and the effects were observed to be persistent for at least several months. Acceptance and mindfulness–based treatments may be more effective than other approaches. Also, patients with musculoskeletal pain may gain more benefits from these treatments than patients with IBS.
Comparing our results to other meta-analytical findings, we identified considerable differences in ESs, which led us to the most important question raised from this research: Do these differences related to cultural variations or they are only the functions of trials’ quality? Although we cannot rule out possible effects of culture, our investigations pointed out to some possible methodological factors which may have elevated the pooled ESs- factors like high risk of bias and small sample sizes. Modifications in the quality of RCTs seem to be necessary to confirm the differences in true ES; and if so, it might be a great opportunity for health care systems in Iran and other similar countries.
Ethical approval
Not applicable.
Competing interests
The authors declared no conflict of interest.
Funding
This research received no financial support.
Authors’ contributions
GF: study design, manuscript revision; MF: study design, manuscript revision; AZ: study design, data collection, statistical analysis and interpretation, and manuscript writing.
Acknowledgment
We wish to thank the anonymous reviewers for their constructive comments.
References
- Turk DC, Wilson HD, Cahana A. Treatment of chronic non-cancer pain. Lancet 2011;377(9784):2226-35. doi: 10.1016/s0140-6736(11)60402-9. [Crossref]
- Demyttenaere K, Bruffaerts R, Lee S, Posada-Villa J, Kovess V, Angermeyer MC, et al. Mental disorders among persons with chronic back or neck pain: results from the World Mental Health Surveys. Pain 2007;129(3):332-42. doi: 10.1016/j.pain.2007.01.022. [Crossref]
- Burke AL, Mathias JL, Denson LA. Psychological functioning of people living with chronic pain: a meta-analytic review. Br J Clin Psychol 2015;54(3):345-60. doi: 10.1111/bjc.12078. [Crossref]
- Mehraban A, Shams J, Moamenzade S, Samimi SM, Rafiee S, Zademohamadi F. The high prevalence of obsessive-compulsive disorder in patients with chronic pain. Iran J Psychiatry 2014;9(4):203-8.
- Naghavi M, Shahraz S, Sepanlou SG, Dicker D, Naghavi P, Pourmalek F, et al. Health transition in Iran toward chronic diseases based on results of Global Burden of Disease 2010. Arch Iran Med 2014;17(5):321-35.
- American Psychiatric Association (APA). Diagnostic and Statistical Manual of Mental Disorders: DSM-5. 5th ed. Arlington, VA: APA; 2013.
- World Health Organization (WHO). Preventing Suicide: A Global Imperative. Vol. 143. Geneva: WHO; 2014.
- Bair MJ, Robinson RL, Katon W, Kroenke K. Depression and pain comorbidity: a literature review. Arch Intern Med 2003;163(20):2433-45. doi: 10.1001/archinte.163.20.2433. [Crossref]
- Goesling J, Clauw DJ, Hassett AL. Pain and depression: an integrative review of neurobiological and psychological factors. Curr Psychiatry Rep 2013;15(12):421. doi: 10.1007/s11920-013-0421-0. [Crossref]
- Edwards RR, Dworkin RH, Sullivan MD, Turk DC, Wasan AD. The role of psychosocial processes in the development and maintenance of chronic Pain. J Pain 2016;17(9 Suppl):T70-92. doi: 10.1016/j.jpain.2016.01.001. [Crossref]
- Knaster P, Karlsson H, Estlander AM, Kalso E. Psychiatric disorders as assessed with SCID in chronic pain patients: the anxiety disorders precede the onset of pain. Gen Hosp Psychiatry 2012;34(1):46-52. doi: 10.1016/j.genhosppsych.2011.09.004. [Crossref]
- Bair MJ, Robinson RL, Eckert GJ, Stang PE, Croghan TW, Kroenke K. Impact of pain on depression treatment response in primary care. Psychosom Med 2004;66(1):17-22. doi: 10.1097/01.psy.0000106883.94059.c5. [Crossref]
- Kroenke K, Shen J, Oxman TE, Williams JW Jr, Dietrich AJ. Impact of pain on the outcomes of depression treatment: results from the RESPECT trial. Pain 2008;134(1-2):209-15. doi: 10.1016/j.pain.2007.09.021. [Crossref]
- 14 Scott EL, Kroenke K, Wu J, Yu Z. Beneficial effects of improvement in depression, pain catastrophizing, and anxiety on pain outcomes: a 12-month longitudinal analysis. J Pain 2016;17(2):215-22. doi: 10.1016/j.jpain.2015.10.011. [Crossref]
- Dworkin RH, Bruehl S, Fillingim RB, Loeser JD, Terman GW, Turk DC. Multidimensional diagnostic criteria for chronic pain: introduction to the ACTTION-American Pain Society Pain Taxonomy (AAPT). J Pain 2016;17(9 Suppl):T1-9. doi: 10.1016/j.jpain.2016.02.010. [Crossref]
- Morley S, Eccleston C, Williams A. Systematic review and meta-analysis of randomized controlled trials of cognitive behaviour therapy and behaviour therapy for chronic pain in adults, excluding headache. Pain 1999;80(1-2):1-13. doi: 10.1016/s0304-3959(98)00255-3. [Crossref]
- Williams AC, Eccleston C, Morley S. Psychological therapies for the management of chronic pain (excluding headache) in adults. Cochrane Database Syst Rev 2012;11(11):CD007407. doi: 10.1002/14651858.CD007407.pub3. [Crossref]
- Hoffman BM, Papas RK, Chatkoff DK, Kerns RD. Meta-analysis of psychological interventions for chronic low back pain. Health Psychol 2007;26(1):1-9. doi: 10.1037/0278-6133.26.1.1. [Crossref]
- Veehof MM, Trompetter HR, Bohlmeijer ET, Schreurs KM. Acceptance- and mindfulness-based interventions for the treatment of chronic pain: a meta-analytic review. Cogn Behav Ther 2016;45(1):5-31. doi: 10.1080/16506073.2015.1098724. [Crossref]
- Hilton L, Hempel S, Ewing BA, Apaydin E, Xenakis L, Newberry S, et al. Mindfulness meditation for chronic pain: systematic review and meta-analysis. Ann Behav Med 2017;51(2):199-213. doi: 10.1007/s12160-016-9844-2. [Crossref]
- Bernardy K, Klose P, Welsch P, Häuser W. Efficacy, acceptability and safety of cognitive behavioural therapies in fibromyalgia syndrome - a systematic review and meta-analysis of randomized controlled trials. Eur J Pain 2018;22(2):242-60. doi: 10.1002/ejp.1121. [Crossref]
- Jensen MP, Turk DC. Contributions of psychology to the understanding and treatment of people with chronic pain: why it matters to ALL psychologists. Am Psychol 2014;69(2):105-18. doi: 10.1037/a0035641. [Crossref]
- Gatchel RJ, Peng YB, Peters ML, Fuchs PN, Turk DC. The biopsychosocial approach to chronic pain: scientific advances and future directions. Psychol Bull 2007;133(4):581-624. doi: 10.1037/0033-2909.133.4.581. [Crossref]
- Pillay T, van Zyl HA, Blackbeard D. Chronic pain perception and cultural experience. Procedia Soc Behav Sci 2014;113:151-60. doi: 10.1016/j.sbspro.2014.01.022. [Crossref]
- Koç V, Kafa G. Cross-cultural research on psychotherapy: the need for a change. J Cross Cult Psychol 2019;50(1):100-15. doi: 10.1177/0022022118806577. [Crossref]
- Kuo BCH. Culture’s consequences on coping: theories, evidences, and dimensionalities. J Cross Cult Psychol 2011;42(6):1084-100. doi: 10.1177/0022022110381126. [Crossref]
- Aldwin CM. Stress, Coping, and Development: An Integrative Perspective. 2nd ed. The Guilford Press; 2007.
- Higgins JP, Altman DG. Assessing risk of bias in included studies. In: Higgins JP, Green S, eds. Cochrane Handbook for Systematic Reviews of Interventions. Chichester, UK: John Wiley & Sons Ltd; 2008. p. 187-241. doi: 10.1002/9780470712184.ch8.
- Deeks JJ, Higgins JP, Altman DG. Analysing data and undertaking meta-analyses. In: Higgins JP, Green S, eds. Cochrane Handbook for Systematic Reviews of Interventions: Cochrane Book Series. Hoboken: Wiley; 2008. doi: 10.1002/9780470712184.ch9.
- Borenstein M. Effect sizes for continuous data. In: Cooper H, Hedges L, Valentine J, eds. The Handbook of Research Synthesis and Meta-Analysis. 2nd ed. Russell Sage Foundation; 2009. p. 221-41.
- Morris SB. Estimating effect sizes from pretest-posttest-control group designs. Organ Res Methods 2008;11(2):364-86. doi: 10.1177/1094428106291059. [Crossref]
- Higgins JP, Deeks JJ, Altman DG. Special topics in statistics. In: Higgins JP, Green S, eds. Cochrane Handbook for Systematic Reviews of Interventions: Cochrane Book Series. Hoboken: Wiley; 2008. doi: 10.1002/9780470712184.ch16.
- Langan D, Higgins JPT, Jackson D, Bowden J, Veroniki AA, Kontopantelis E, et al. A comparison of heterogeneity variance estimators in simulated random-effects meta-analyses. Res Synth Methods 2019;10(1):83-98. doi: 10.1002/jrsm.1316. [Crossref]
- Viechtbauer W, Cheung MW. Outlier and influence diagnostics for meta-analysis. Res Synth Methods 2010;1(2):112-25. doi: 10.1002/jrsm.11. [Crossref]
- Jin ZC, Zhou XH, He J. Statistical methods for dealing with publication bias in meta-analysis. Stat Med 2015;34(2):343-60. doi: 10.1002/sim.6342. [Crossref]
- Viechtbauer W. Conducting meta-analyses in R with the metafor package. J Stat Softw 2010;36(3):1-48. doi: 10.18637/jss.v036.i03. [Crossref]
- Del Re AC. Compute.es: compute effect sizes. R Package version 0.2. 2013. Available from: http://cran.r-project.org/web/packages/compute.es. Accessed June 29, 2020.
- Borenstein M, Hedges LV, Higgins JP, Rothstein HR. Introduction to Meta-Analysis. John Wiley & Sons Ltd; 2009. doi: 10.1002/9780470743386.
- Wood L, Egger M, Gluud LL, Schulz KF, Jüni P, Altman DG, et al. Empirical evidence of bias in treatment effect estimates in controlled trials with different interventions and outcomes: meta-epidemiological study. BMJ 2008;336(7644):601-5. doi: 10.1136/bmj.39465.451748.AD. [Crossref]
- Kühberger A, Fritz A, Scherndl T. Publication bias in psychology: a diagnosis based on the correlation between effect size and sample size. PLoS One 2014;9(9):e105825. doi: 10.1371/journal.pone.0105825. [Crossref]
- Alighias M. Design and Evaluation of Mental Health Training Package with Chronic Back Pain in Nurses Working in Hospitals Valieasr [dissertation]. Tehran: Tarbiat Modares; 2015. [Persian].
- Alighias M, Tavafian SS, Niknami S. Effect of interventional education based on psychological factors among nurses suffering from chronic low back pain: a randomized clinical trial. Toloo-E-Behdasht 2016;14(6):384-95. [Persian].
- Anvari M, Ebrahimi A, Neshat Doost HT, Afshar H. The effectiveness of group-based acceptance and commitment therapy on pain-related anxiety, acceptance of pain and pain intensity in patients with chronic pain. Journal of Isfahan Medical School 2014;32(295):1156-65. [Persian].
- Arvand J, Sakhayi A, Jadidi H. Efficacy of music therapy on anxiety and diarrhea among patients with irritable bowel syndrome. Medical Sciences Journal 2017;27(3):194-200. [Persian].
- Asadolahi F. Comparison of the effects of mindfulness based cognitive therapy and spiritual therapy on somatic symptoms, anxiety and depression in female patients with irritable bowel syndrome [dissertation]. Isfahan: University of Isfahan; 2013. [Persian].
- Esmailpoor B, Aleyasin SA. Effectiveness of emotional intelligence training on anxiety and quality of life for patients with irritable bowel syndrome. Journal Psychology and Educational Sciences 2017;5(2):45–60. [Persian].
- Esmailpoor B, Aleyasin SA. Effectiveness of emotional intelligence training on quality of life for patients with irritable bowel syndrome. Journal of Psychological Models and Methods 2017;7(6):105-20. [Persian].
- Adarvishi S, Asadi M, Zarea K, GhasemiDeh Cheshmeh M, Ahmadnea M, Hardani F. Effect of problem solving training on anxiety in patients with IBS. Preventive Care in Nursing and Midwifery Journal 2016;5(2):22-32. [Persian].
- Foroudastan M. Comparing the efficacy of cognitive-behavioral therapy, quality of life therapy and massage therapy on the intensity of pain, quality of pain, depression, anxiety and stress in women with chronic musculoskeletal pain [dissertation]. Isfahan: University of Isfahan; 2013. [Persian].
- Pourmohseni-Koluri F, Eslami F. The effectiveness of group cognitive therapy based on mindfulness on mental health and quality of life of patients with irritable bowel syndrome. Quarterly Journal of Health Psychology 2017;5(20):48-67. [Persian].
- Golchin N. The Efficacy of Cognitive Behavior Therapy on Improving of Pain and Enhancing Mental Health in Chronic Back Pain Individuals [dissertation]. Tehran: Payame Noor; 2010. [Persian].
- Jonbozorgi M, Golchin N, Alipour A, Agah Heris M. The effectiveness of group cognitive-behavior therapy on decreasing severity of pain and psychological distress among women with chronic back pain. Iranian Journal of Psychiatry and Clinical Psychology 2013;19(2):102-8. [Persian].
- Hazrati M, Hoseini M, Dejbakhsh T, Taghavi SA, Rajaeefard A. The effect of Benson relaxation therapy on anxiety level and severity of symptoms in patients with irritable bowel syndrome. Journal of Arak University of Medical Sciences 2006;9(4):9-17. [Persian].
- Hoseini M, Nikrooz L, Poursamad A, Taghavi A, Rajaeefard A. The effect of Benson relaxation technique on anxiety level and qquality of life of patients with irritable bowel syndrome. Armaghane Danesh 2009;14(1):101-11. [Persian].
- Haghayegh SA, Kalantari M, Molavi H, Talebi M. Cognitive-behavior therapy in patients with irritable bowel syndrome. J Iran Psychol 2008;4(16):377-86. [Persian].
- Irandoost F, Safari S, Neshat Doost HT, Nadi MA. The effectiveness of Group Acceptance and Commitment Therapy (ACT) on pain related anxiety and depression in women with chronic low back pain. Int J Behav Sci 2015;9(1):1.
- Jokar S. The effectiveness of cognitive-behavioral stress management on depression and anxiety and stress symptoms, and the improvement of health status in patients with rheumatoid arthritis [dissertation]. Isfahan: University of Isfahan; 2009. [Persian].
- Khoshsorour S. Investigating the effect of neurofeedback in decreasing anxiety and symptom severity of patients with irritable bowel syndrome (IBS). The Journal of Urmia University of Medical Sciences 2018;28(10):647-58. [Persian].
- Soltanian G. The effectiveness of cognitive-behavioral group therapy on quality of life, depression, anxiety and stress in patients with rheumatoid arthritis [dissertation]. Semnan: University of Semnan; 2014. [Persian].
- Sabour S, Kakabraee K. The effectiveness of acceptance and commitment therapy on depression, stress and indicators of pain in women with chronic pain. Iranian Journal of Rehabilitation Research in Nursing 2016;2(4):1-9. doi: 10.21859/ijrn-02041. [Persian]. [Crossref]
- Tabatabaee Shahrabad A. The Effectiveness of Metacognitive Therapy on Anxiety, Depression and Quality of life of Patients with Irritable Bowel Syndrome [dissertation]. Mashhad: Ferdowsi University; 2014. [Persian].
- Kamkar A. The Effectiveness of group Cognitive – Behavioral Stress Management on physical Symptoms of Patients with Irritable Bowel Syndrome With and Without Comorbidity of Anxiety and Depression and on Increase of Their Quality of Life [dissertation]. Tehran: Allameh Tabataba’i University; 2011. [Persian].
- Masumian S. The Effect of Mindfulness-Based Stress Reduction (MBSR) on Decreasing Depression, increasing Quality of Life and self-efficacy of the Chronic Low back Pain Patients (CLBP) [dissertation]. Tehran: Allameh Tabataba’i University; 2013. [Persian].
- Masumian S, Golzari M, Shairi MR, Momenzadeh S. The effect of mindfulness-based stress reduction on the rate of depression in patients with chronic low back pain. Journal of Anesthesiology and Pain 2013;3(2):82-8. [Persian].
- Rahimian Boogar I, Tabatabaeian M. Effect of cognitive-behavioral group therapy on depression of the patients with chronic low back pain: a 4-months follow up. Koomesh 2012;13(2):209-17. [Persian].
- Salayani F. Comparison of effectiveness of cognitive group therapy and combination it with self-management program in reducing of pain, depression, anxiety, aggression in women with chronic pain [dissertation]. Mashhad: Ferdowsi University; 2015. [Persian].
- Asghari EbrahimAbad M, Salayani F, Mashhadi A, Attarzadeh Hosseini R, Mashhadi Nejad H, Eftekharzadeh S, et al. The effectiveness of cognitive group therapy on anxiety andaggression of females with chronic pain. Anesthesiology and Pain 2015;6(1):34-44. [Persian].
- Shojaei Z. The Effectiveness of transdisciplinary training on improving signs of depression and adaptation in patients with chronic pain [dissertation]. Shahrood: Azad Islamic University-Shahrood Branch; 2016.[Persian].
- Mohamadi J, Ghazanfari F, Mir Drikvand F. Efficacy of mindfulness-based cognitive therapy on anxiety and depression in irritable bowel syndrome. Community Health Journal 2017;11(3-4):11-9. doi: 10.22123/chj.2018.102588.1052. [Persian]. [Crossref]
- Mahvi-Shirazi M. The mental health and personality traits of IBS patients in comparison with IBD patients and healthy persons and also survey of effectiveness of medical therapy and cognitive behavioral therapy for treatment of IBS patients [dissertation]. Tehran: Tarbiat Modares University; 2008. [Persian].
- Mahvi-Shirazi M, Fathi-Ashtiani A, Rasoolzade-Tabatabaei SK, Amini M. Irritable bowel syndrome treatment: cognitive behavioral therapy versus medical treatment. Arch Med Sci 2012;8(1):123-9. doi: 10.5114/aoms.2012.27292. [Crossref]
- Vakili N. Effectiveness of group cognitive behavior therapy on depression, quality of life of patients with low back pain. [dissertation]. Isfahan: University of Isfahan; 2009. [Persian].
- Najafi MR, Vakili N, Neshat Doost HT, Asgari K, Rezaei F. The effect of cognitive-behavioral group pain management therapy on depression of the female with chronic low back pain. J Clin Psychol 2009;1(4):11-9. doi: 10.22075/jcp.2017.1983. [Persian]. [Crossref]
- Yeganeh S. Investigate effectiveness of mindfulness based stress reduction on the stress (MBSR), anxiety, depression and psychological well being in patients with systemic lupus erythematosus and rheumatoid arthritis [dissertation]. Bandar Abbas: Islamic Azad University-Bandar Abbas Branch; 2015. [Persian].
- Yousefzadeh M, Talebi H, Neshat Doost HT. The effect of schema therapy on maladaptive schemas in Tehranian patients with chronic low back pain: a two months follow-up. Research in Cognitive and Behavioral Sciences 2018;7(2):69-82. doi: 10.22108/cbs.2017.100397.0. [Persian]. [Crossref]
- Yousefzadeh M, Neshat Doost HT, Talebi H. A comparison of the effectiveness of cognitive-behavior therapy and schema therapy on the reduction of depression among patients with chronic low back pain in Tehran, Iran. Journal of Research in Behavioural Sciences 2017;15(1):67-77. [Persian].
- Mo’tamedi H, Rezaiemaram P, Tavallaie A. The effectiveness of a group-based acceptance and commitment additive therapy on rehabilitation of female outpatients with chronic headache: preliminary findings reducing 3 dimensions of headache impact. Headache 2012;52(7):1106-19. doi: 10.1111/j.1526-4610.2012.02192.x. [Crossref]
- Kafi M, Afshar H, Moghtadaei K, Ariapooran S, Daghaghzadeh H, Salamat M. Effectiveness of mindfulness-based cognitive-therapy on psychological signs women with irritable bowel syndrome. Koomesh 2014;15(2):255-64. [Persian].
- Moghtadaei K. Effectiveness of mindfulness-based cognitive therapy on psychological signs and somatic symptoms in patients with irritable bowel syndrome [dissertation]. Gilan: University of Gilan; 2012. [Persian].
- Nazemi Ardakani F, Bahrami Ehsan H, Alipour A, Bayat N. Effectiveness of religious - spiritual intervention based on Islamic approach on psychological and immune indices in patients with Rheumatoid Arthritis. Journal of Applied Psycology Research 2016;6(4):113-31. doi: 10.22059/japr.2016.57967. [Persian]. [Crossref]
- Nazemi Ardakani F, Bahrami Ehsan H, Alipour A, Bayat N. Effectiveness of cognitive behavioral stress management intervention on psychological and immune factors in patients with Rheumatoid Arthritis referred to the rheumatology clinics of Baghiat Allah hospital. Journal of Military Psychology 2015;6(22):55-65. [Persian].
- Haugmark T, Hagen KB, Smedslund G, Zangi HA. Mindfulness- and acceptance-based interventions for patients with fibromyalgia-a systematic review and meta-analyses. PLoS One 2019;14(9):e0221897. doi: 10.1371/journal.pone.0221897. [Crossref]
- Dixon KE, Keefe FJ, Scipio CD, Perri LM, Abernethy AP. Psychological interventions for arthritis pain management in adults: a meta-analysis. Health Psychol 2007;26(3):241-50. doi: 10.1037/0278-6133.26.3.241. [Crossref]